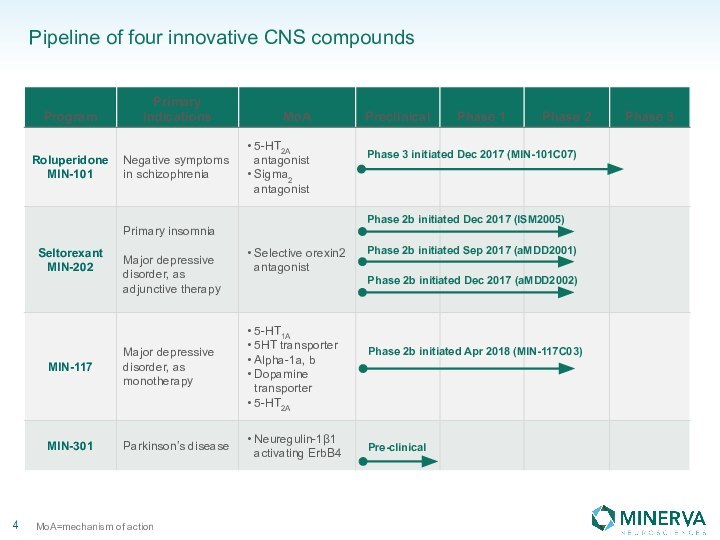
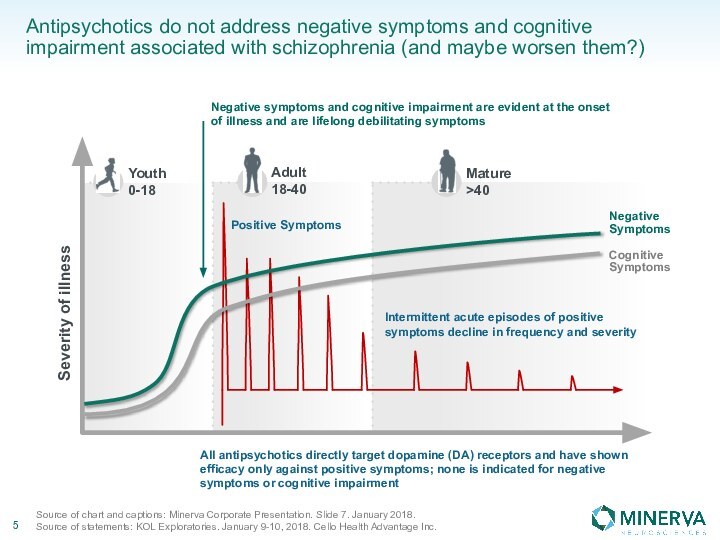
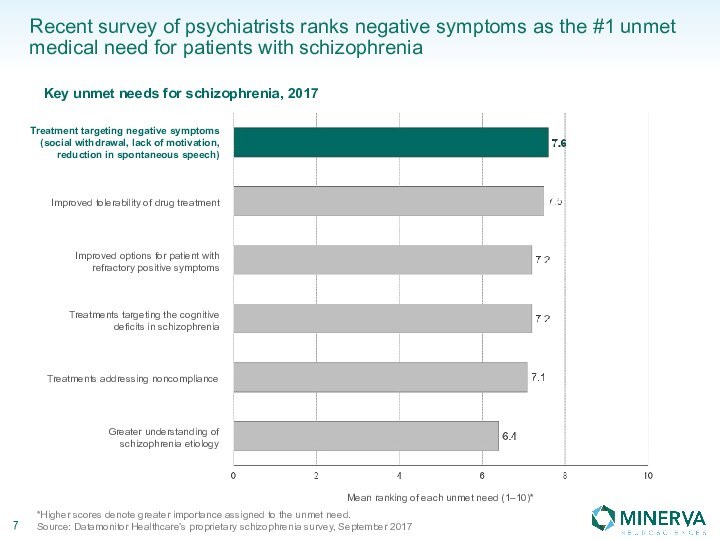
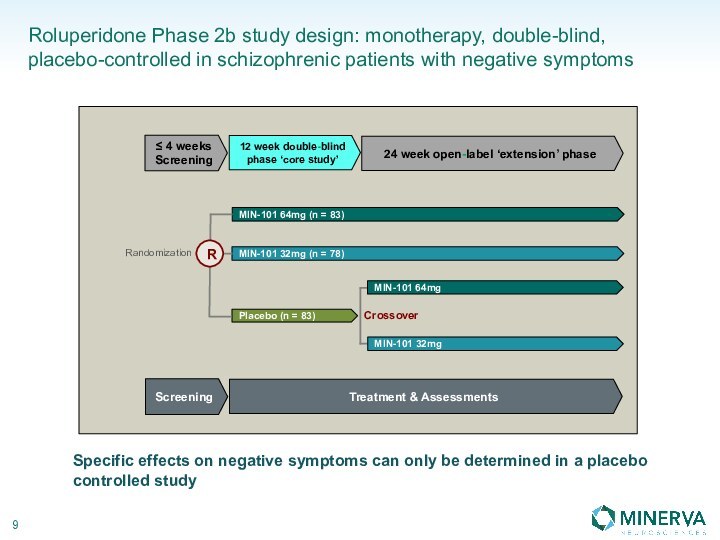
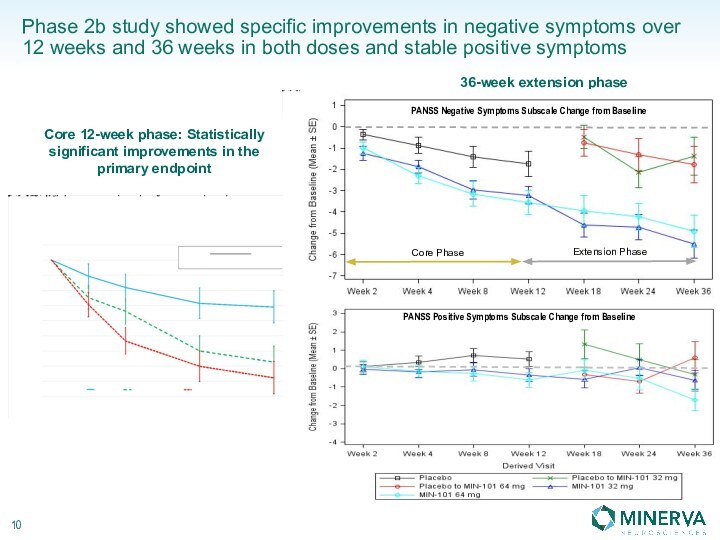
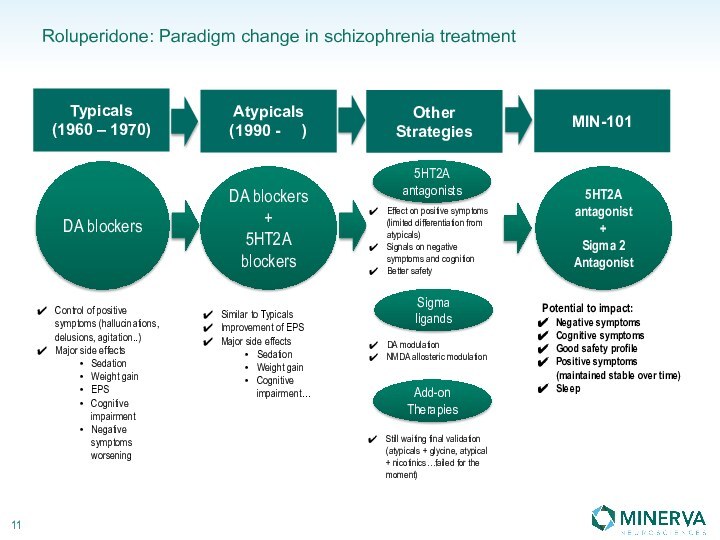
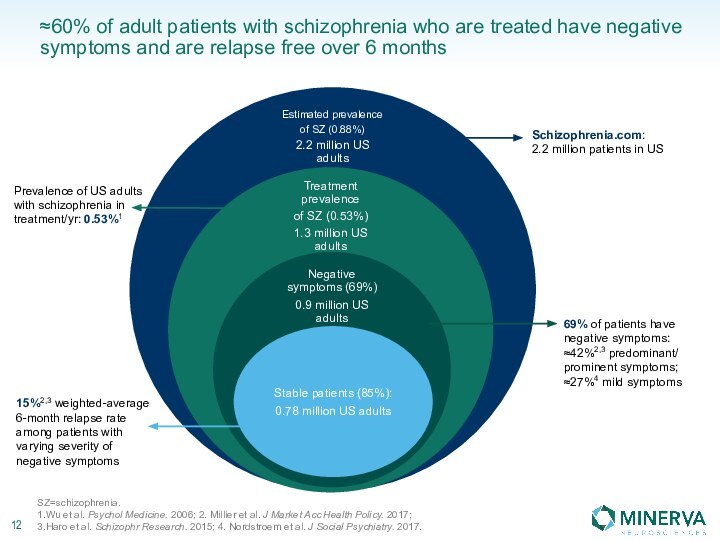
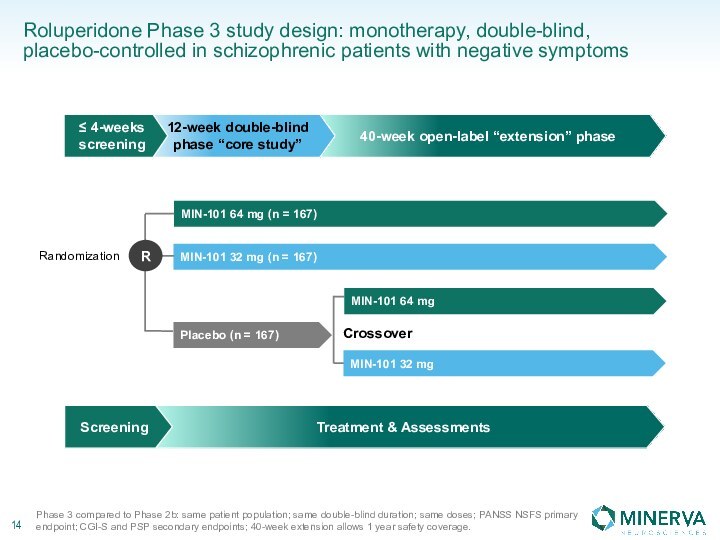
in this presentation are the property of their respective
owners.Forward-Looking Statement Safe-Harbor
This presentation contains forward-looking statements about Minerva Neurosciences which are subject to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995, as amended. Forward-looking statements are statements that are not historical facts, reflect management’s expectations as of the date of this presentation, and involve certain risks and uncertainties. Forward-looking statements include, but are not limited to: the benefits, efficacy and safety of our new formulations; the potential of the diagnosis and treatment of negative symptoms of schizophrenia and other diseases; whether studies performed on analogs or backups of our compounds are a good predictor of the clinical efficacy of our compounds; statements with respect to the timing and results of future clinical milestones with roluperidone (MIN-101), seltorexant (MIN-202) and MIN-117, including the Phase 3 trial of roluperidone, the Phase 2b trials of seltorexant and the Phase 2b trial of MIN-117; statements regarding our ability to successfully develop and commercialize our therapeutic products; our expectations regarding approval for our products by the U.S. Food and Drug Administration or equivalent foreign regulatory agencies; estimates regarding the market potential for our products; and future performance. All of such statements are subject to certain risks and uncertainties, many of which are difficult to predict and generally beyond the control of the Company, that could cause actual results to differ materially from those expressed in, or implied or projected by, the forward-looking statements. These forward-looking statements are based on our current expectations and may differ materially from actual results due to a variety of factors including, without limitation, whether any of our therapeutic products will advance further in the clinical trials process and whether and when, if at all, they will receive final approval from the U.S. Food and Drug Administration or equivalent foreign regulatory agencies and for which indications; whether the results of future clinical trials of roluperidone, seltorexant, MIN-117 and MIN-301, if any, will be consistent with the results of past clinical trials; whether roluperidone, seltorexant, MIN-117 and MIN-301 will be successfully marketed if approved; whether our therapeutic product discovery and development efforts will be successful; our ability to achieve the results contemplated by our co-development agreements; the strength and enforceability of our intellectual property rights; competition from pharmaceutical and biotechnology companies; the development of and our ability to take advantage of the market for our therapeutic products; our ability to raise additional capital to fund our operations on terms acceptable to us; and general economic conditions. These and other potential risks and uncertainties that could cause actual results to differ from the results predicted are more fully detailed under the caption “Risk Factors” in our filings with the Securities and Exchange Commission, including our Quarterly Report on Form 10-Q for the quarter ended March 31, 2018, filed with the Securities and Exchange Commission on May 3, 2018, as well as our Annual Report on Form 10-K for the year ended December 31, 2017, filed with the Securities and Exchange Commission on March 12, 2018. Copies of reports filed with the SEC are posted on our website at www.minervaneurosciences.com. Our audience is cautioned not to place undue reliance on these forward-looking statements that speak only as of the date hereof, and we disclaim any obligation to update any forward-looking statements, except as required by law.