- Главная
- Разное
- Бизнес и предпринимательство
- Образование
- Развлечения
- Государство
- Спорт
- Графика
- Культурология
- Еда и кулинария
- Лингвистика
- Религиоведение
- Черчение
- Физкультура
- ИЗО
- Психология
- Социология
- Английский язык
- Астрономия
- Алгебра
- Биология
- География
- Геометрия
- Детские презентации
- Информатика
- История
- Литература
- Маркетинг
- Математика
- Медицина
- Менеджмент
- Музыка
- МХК
- Немецкий язык
- ОБЖ
- Обществознание
- Окружающий мир
- Педагогика
- Русский язык
- Технология
- Физика
- Философия
- Химия
- Шаблоны, картинки для презентаций
- Экология
- Экономика
- Юриспруденция
Что такое findslide.org?
FindSlide.org - это сайт презентаций, докладов, шаблонов в формате PowerPoint.
Обратная связь
Email: Нажмите что бы посмотреть
Презентация на тему Pharmaceutical monitoring and evaluation
Содержание
- 2. TopicsConcepts on pharmaceutical assessment/monitoring The WHO process
- 3. Pharmaceutical monitoring/ evaluationMonitoringReview of the progress re
- 4. Who can use the results from assessment
- 5. Develop implementation plans and identify strategies &
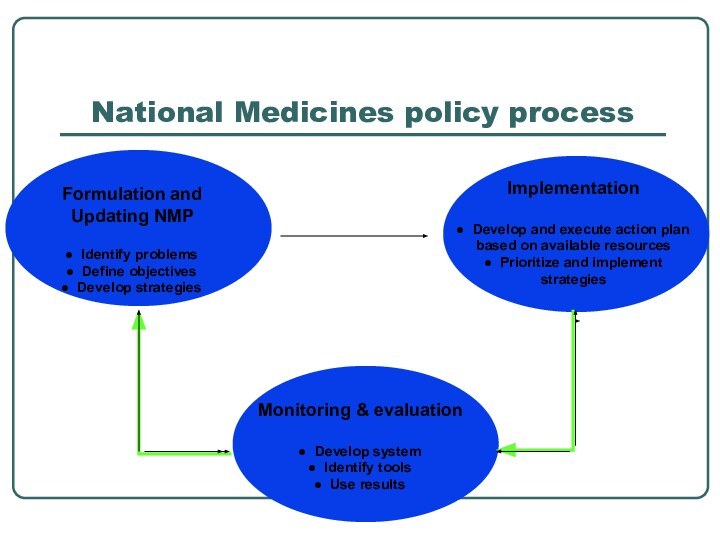
- 6. National Medicines policy process
- 7. WHO hierarchical approach to monitoring and assessing
- 8. Level I indicators: structure and process indicatorsRegular
- 9. Level II- facility outcome and impactindicators: WHO
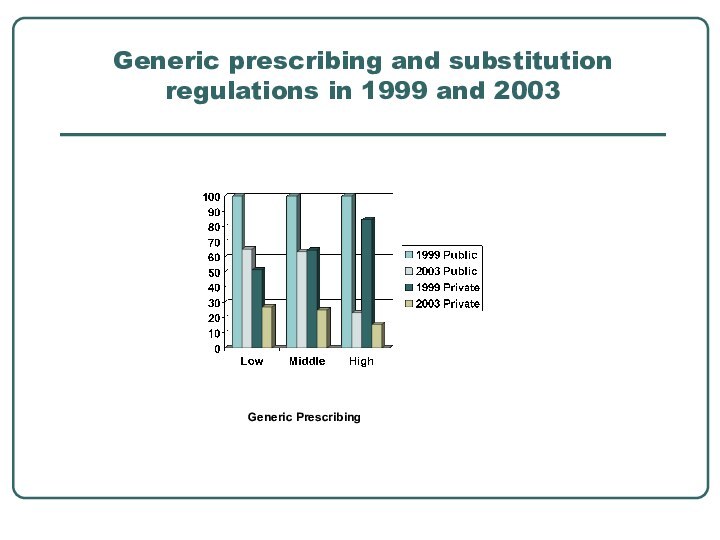
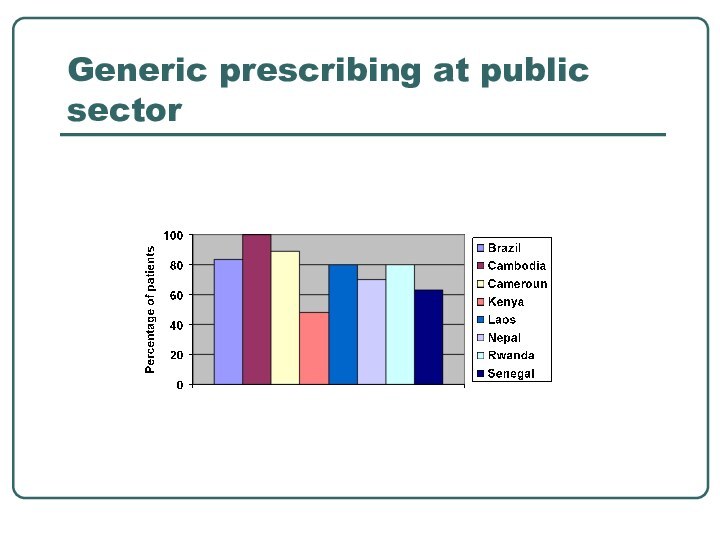
- 10. Generic prescribing and substitution regulations in 1999 and 2003 Generic Prescribing
- 11. Generic prescribing at public sector
- 12. Measuring access to essential medicines ( Household
- 13. Importance of household survey Household situations How
- 14. Indicators: (few examples)AffordabilityAverage household medicine expenditures as
- 15. Indicators: (few examples)Rational Use of MedicinesPercent of
- 16. Current issues on household survey processChallenge to
- 17. Level III IndicatorsSystematic survey and monitoringDrug price
- 18. Sampling issues for systematic surveyFollow specific procedures
- 19. Sampling Recommendation for Level II facility surveySampling
- 20. The household survey sampling scheme (non probability,
- 21. Is the sampling frame valid? (clustering in
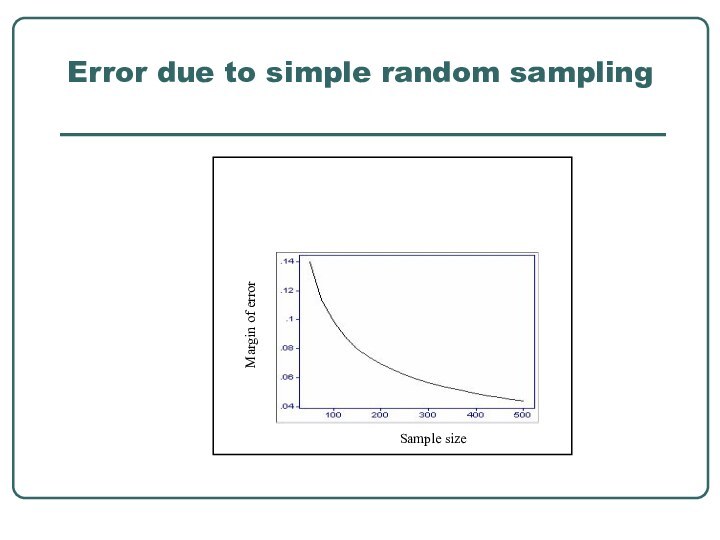
- 22. Error due to simple random sampling
- 23. Who can be trained to do the
- 24. Preparing and implementing systematic survey Administrative preparation:
- 25. Pharmaceutical indicatorsVariables that measure situations and changeNumerical
- 26. Why is it important to use indicators?Standard
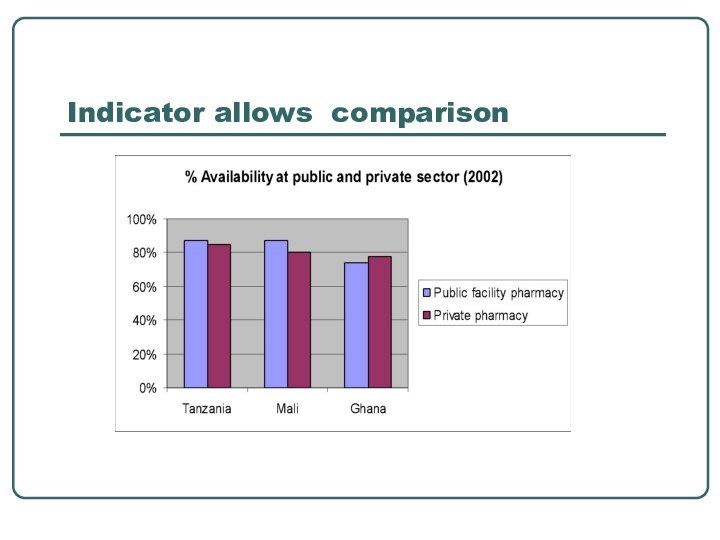
- 27. Indicator allows comparison
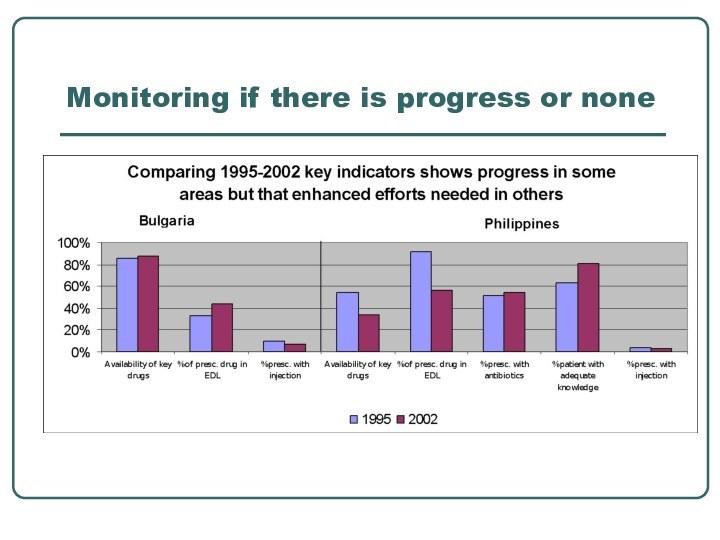
- 28. Monitoring if there is progress or none
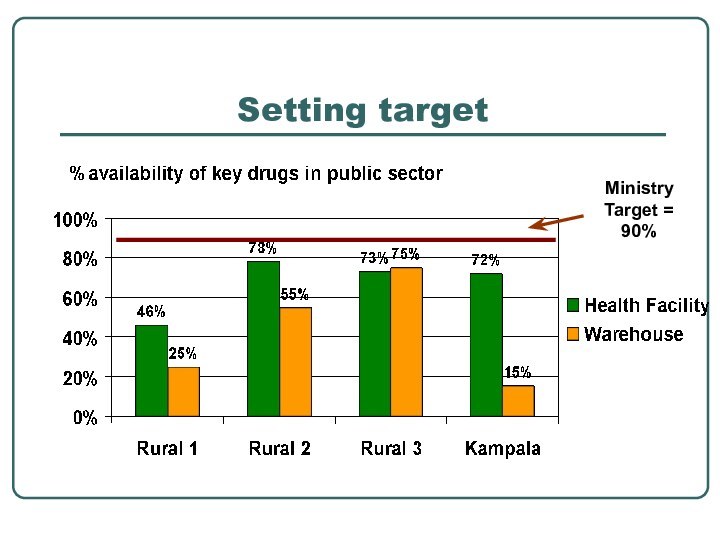
- 29. Setting target
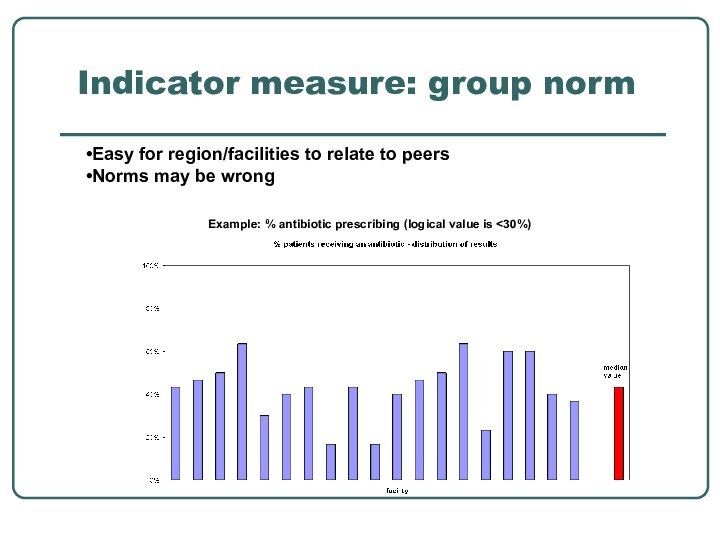
- 30. Indicator measure: group norm Example: % antibiotic prescribing (logical value is
- 31. Summarizing indicator measuresPercentage: yes or no over
- 32. Indicator measure: Ideal/logical valuesLogical value exist for
- 33. Connecting Survey Results and Interventions
- 34. The way forward on country monitoringEvidence through
- 35. Скачать презентацию
- 36. Похожие презентации

























Слайд 2
Topics
Concepts on pharmaceutical assessment/monitoring
The WHO process on
assessing and monitoring pharmaceutical situation
concepts on indicators
Слайд 3
Pharmaceutical monitoring/ evaluation
Monitoring
Review of the progress re completion,
allows for corrective action, focus on inputs and outputs
Common
methods Supervisory visits
Routine reporting of selected data
Sentinel sites for more detailed reporting/ intensive monitoring
Special studies for specific additional information
Evaluation
Part of overall pharmaceutical assessment, progress on meeting objectives
Types of evaluations
Needs assessment (situation analysis,
Formative evaluation (midterm review)
Summative evaluation (final evaluation)
Field surveys using standard pharmaceutical indicators & ongoing monitoring system, document review
Strategies developed in parallel for comprehensive unified strategy
Слайд 4 Who can use the results from assessment and
monitoring?
Countries - focus action, prioritize, measure achievement
National policy-makers
synchronise policies
data and information to donors and other governmental agencies
International agencies
to assess the structure and capability of countries, assess the progress, accomplishment and impact of aid
Professional groups, NGOs and academia
to focus advocacy activities and information campaigns
Health facilities to be aware of institutional problems & improve situations
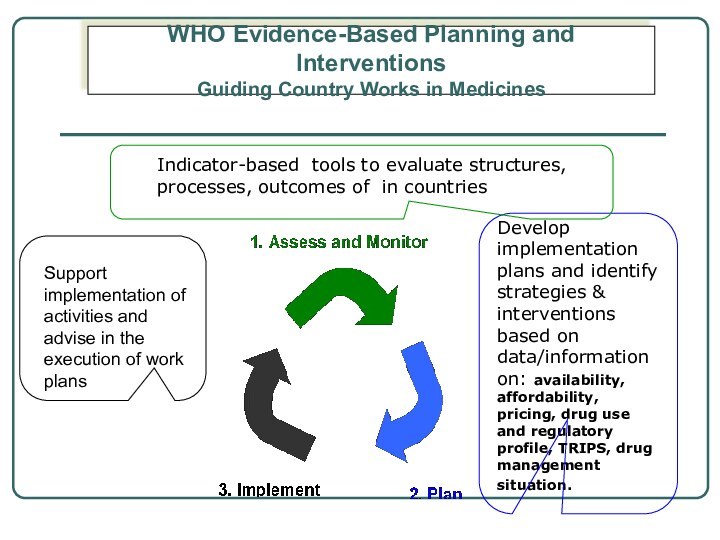
Слайд 5 Develop implementation plans and identify strategies & interventions
based on data/information on: availability, affordability, pricing, drug use
and regulatory profile, TRIPS, drug management situation.Support implementation of activities and advise in the execution of work plans
Indicator-based tools to evaluate structures, processes, outcomes of in countries
WHO Evidence-Based Planning and Interventions
Guiding Country Works in Medicines
Слайд 7 WHO hierarchical approach to monitoring and assessing pharmaceutical
situations
Level I
Core structure
& process indicators
Level II
Core outcome/impact indicators
& household survey
Level III
Indicator tools for specific components
of the pharmaceutical sector
Pricing ● Traditional medicine
HIV/AIDS ● Assessing regulatory capacity
TRIPS
Systematic
survey
Questionnaire
(Health Officials)
Level I
Questionnaire/rapid assessment/checklist
Arrays achievement & weaknessess, illustrate sectoral approaches
Level II
Comprehensive monitoring of pharmaceutical strategy outcome and impact
Measures attainment of objectives
Level III
More detailed indicators for monitoring and evaluating specific areas/components
Слайд 8
Level I indicators: structure and process indicators
Regular survey
questionnaire
Inexpensive process to get information across countries
Can be done
repeatedly/regular periodAutomated questionnaire and data encoding processing
Contents
National Medicines Policy
Regulatory system (marketing authorization, licensing, regulatory inspection, etc)
Medicines supply system, medicines financing, production and trade
Rational use of drugs
Слайд 9 Level II- facility outcome and impactindicators: WHO Operational
Package for Monitoring and Assessing County Pharmaceutical Situations"
Sytematic survey
Indicators
on availability, stock out, record keeping and expiry of key drugs
conservation conditions and handling of medicines
affordability (child and adult moderate pneumonia and option for other disease condition
drug prescribing, dispensing, patient knowledge
practical/operational system of managing a systematic survey and resources
17 survey forms-public health facilities, public pharmacy/dispensary, private pharmacy, warehouses
manual calculation and automated system for descriptive analysis
Слайд 12
Measuring access to essential medicines ( Household Survey)
Level
I and Level II- facility surveys do not measure
access from the patient/consumer perspective.Only household surveys can provide population-based information about how pharmaceutical policies affect the well-being of individuals.
Слайд 13
Importance of household survey
Household situations
How they
access their medicines, where they get them
How much they
payIdentify access and affordability in relation to socio economic indicators, barriers
Examine use of medicines (acute and chronic diseases)
Perceptions on access, use and quality; handling of medicines
Слайд 14
Indicators: (few examples)
Affordability
Average household medicine expenditures as %
of total/non-food/health expenditures
Average household medicine expenditures for a reported
illness (acute, chronic, by illness)% of households with at least partial medicine insurance coverage
Mixed Indicators of Access (availability)
Percent of households reporting a serious acute illness who sought care outside but did not take any medicine.
Percent of households who do not have at home a medicine prescribed to a chronically ill person.
Слайд 15
Indicators: (few examples)
Rational Use of Medicines
Percent of antibiotics
kept for future use
Percent of household medicines with adequate
label/ adequate primary packagingPerception of quality
Percent of respondents who agree that quality of services at their public health care facility is good / quality of services by private provider is good
Percent of respondents who agree that brand name medicines are better than generics/ imported medicines are of better quality than locally manufactured medicines.
Слайд 16
Current issues on household survey process
Challenge to use
population based data to policy evaluation, development and planning
Segregation by socio economic profile
No basic guideline standard???on household survey
What is a household / who is a household member
Sampling
Recall periods- ( number of days, self report, caregivers)
Type of survey (general population, disease based survey)
Слайд 17
Level III Indicators
Systematic survey and monitoring
Drug price survey
and monitoring
WHO/INRUD RDU indicators
Rapid assessment
Global survey on Paediatrics medicines
Questionnaire
on public sector medicines procurement and supply management systems in countriesAssessment of regulatory capacity
Слайд 18
Sampling issues for systematic survey
Follow specific procedures to
minimize selection bias and is representative of the reference
populationA balance between what is desirable and what is feasible- smallest one with a degree of precision
Слайд 19
Sampling Recommendation for Level II facility survey
Sampling (stratification,
random)
5 regions/districts
1 should be among the lowest income
generating areas1 should be the largest or capital city
3 others should be randomly selected
30 facilities each
30 cases per facility
Systematic sampling
Non probability / purposive/ quota sampling
Слайд 20
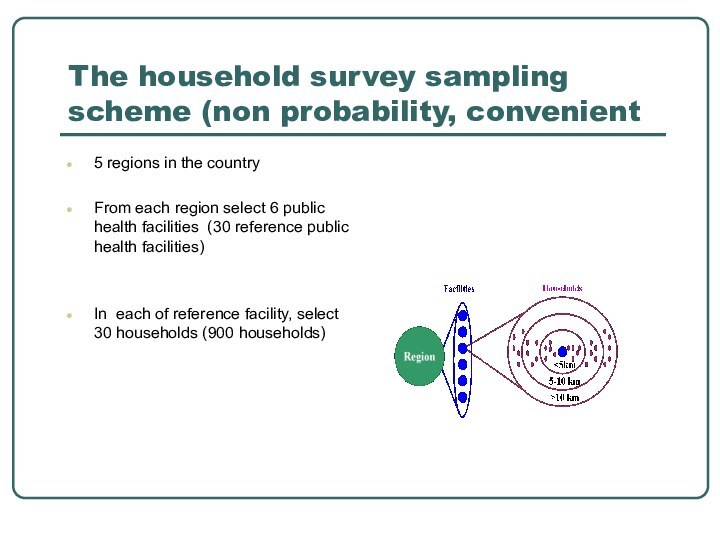
The household survey sampling scheme (non probability, convenient
5 regions in the country
From each region select 6
public health facilities (30 reference public health facilities)In each of reference facility, select 30 households (900 households)
Слайд 21 Is the sampling frame valid? (clustering in drug
supply or drug use data)
Geographic Characteristics
Administration and drug supply
systemEpidemiologic or socio-economic differences
Health Facility Characteristics
Differences in management
Peer norms and collective habits
Provider Characteristics
Training, knowledge, clinical experience
Economic incentives
Industry pressure
Result: Effective sample size is reduced
Слайд 23
Who can be trained to do the survey?
Physicians,
nurses, pharmacists or paramedical staff
Health ministry/department staff and temporary
employees (health related background and experience) data collectors from different parts of the country (language differences)
Слайд 24
Preparing and implementing systematic survey
Administrative preparation:
Coordinating
with WHO, ministry/department of health, public health facilities, private
drug outlets, warehousesMaking logistic arrangements and budget allocations
Technical requirements:
Tailoring the tool-specific items of the survey forms, e.g. key basket of medicines, treatment guidelines, etc.
Training data collectors to carry out the survey and use the survey and summary forms
Analyzing and computing the data
Preparing a report and using result
Слайд 25
Pharmaceutical indicators
Variables that measure situations and change
Numerical (
numbers, percentage, or averages)
Binomials (yes” and “no)”
Linked
to an important input, process, or outcomeWell-established indicators can be adapted/ modified to reflect the realities
Field test
Слайд 26
Why is it important to use indicators?
Standard indicators
facilitates:
comparing the performance of facilities, districts, urban vs rural,
private & public sector, overall situations in countriesseeing trends over time
setting target
Слайд 30
Indicator measure: group norm
Example: % antibiotic prescribing
(logical value is
peersNorms may be wrong
Слайд 31
Summarizing indicator measures
Percentage: yes or no over total
Measures of central tendency
Mean: average value, sensitive to outliers,
weighed toward skewed value, best summary of normally distributed valuesMedian: middle value, resistant to outliers, good summary of any distribution
Equivalent if data are normally distributed
Measure of variation
25th and 75th percentiles: boundaries of middle half of values, good summary of the overall spread of values, better summary of skewed data
Слайд 32
Indicator measure: Ideal/logical values
Logical value exist for some
Logical
value (100%-adequate labelling, meds dispensed, adherence to STG, availability
of medicines, generic, adequacy of storage; 0 days of stock out,)Others need further studies
affordability ( economic profile)
Antibiotic use and injection, meds prescribes are more complex- are (<30, <20 and < 2 and can be controversial)
Optimal value largely depend on disease pattern, policies and treatment G/L and vary from country to country
These values can be calculated empirically
Слайд 34
The way forward on country monitoring
Evidence through systematic
but feasible data collection process is necessary in policy
making and activity implementation. This should include population based informationShould demonstrate that in the long run regular monitoring and evaluation is not difficult and can be done in a cost efficient manner
Portion of country support budget and project grants should be allotted to monitoring and evaluation using indicators
Timely report and information/data sharing